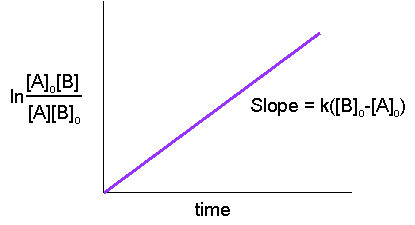
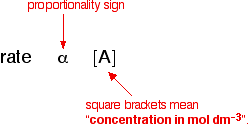
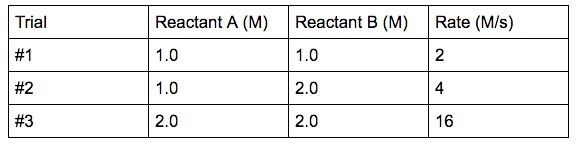
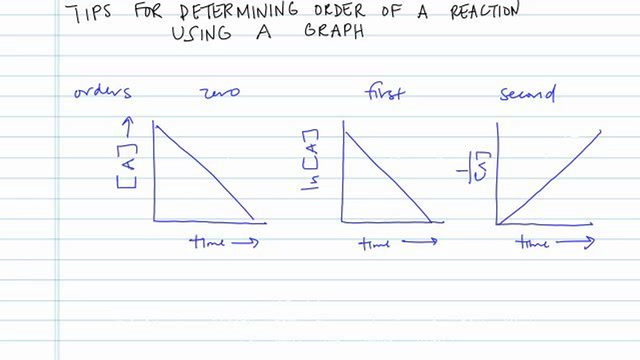
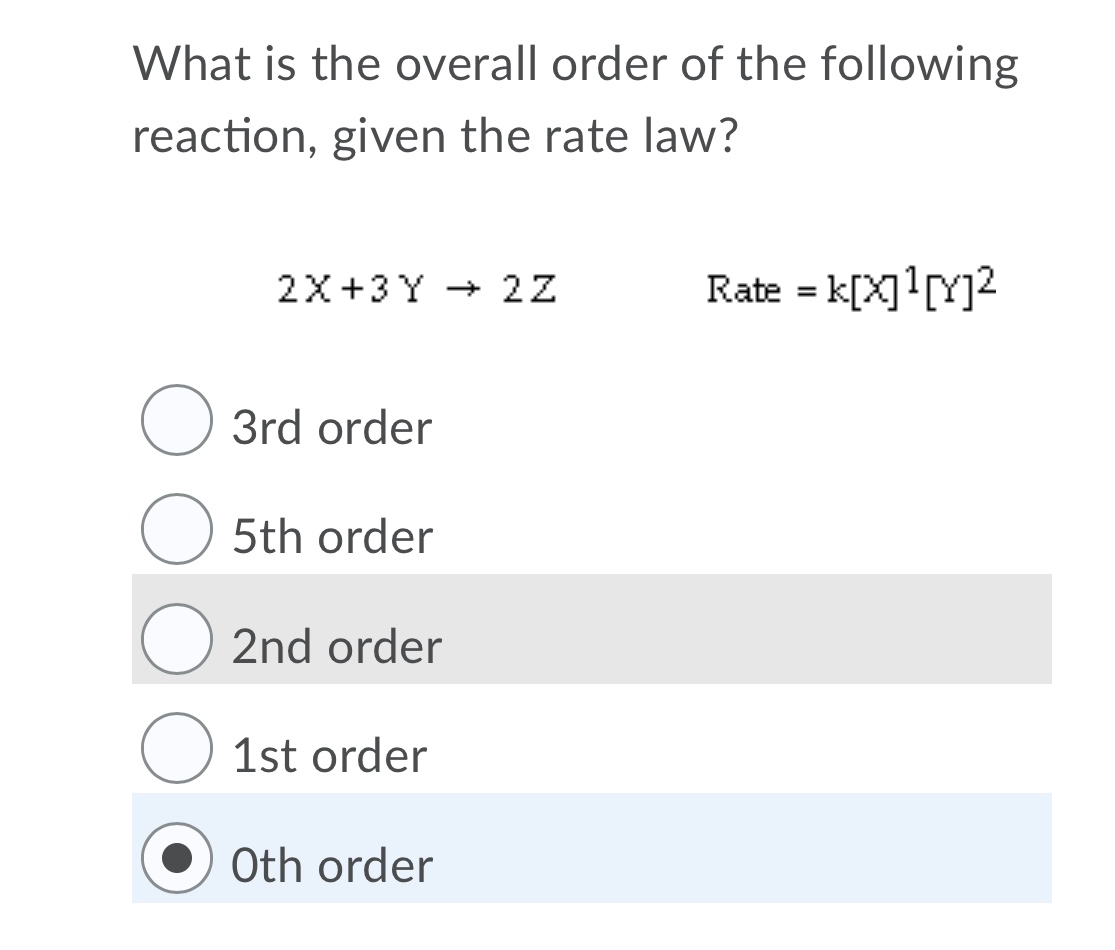
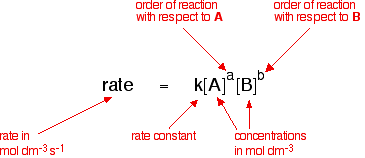
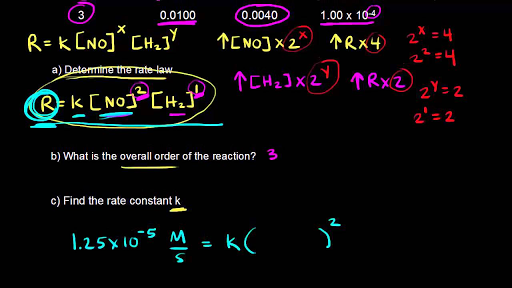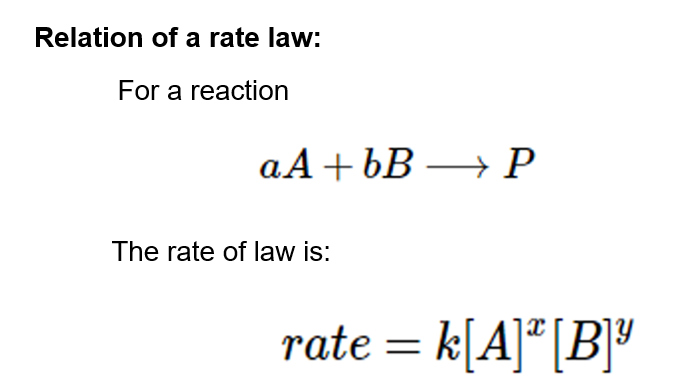Collision Theory For a rx to occur reacting molecules must collide so 1 - The old bonds “loosen up”, or start to break apart 2 - The correct orientation. - ppt download

1. Kinetics 1.1 The rate of chemical reactions 1.2 The rate expression and order of reaction 1.3 Determining the rate equation 1.4 The rate determining. - ppt download